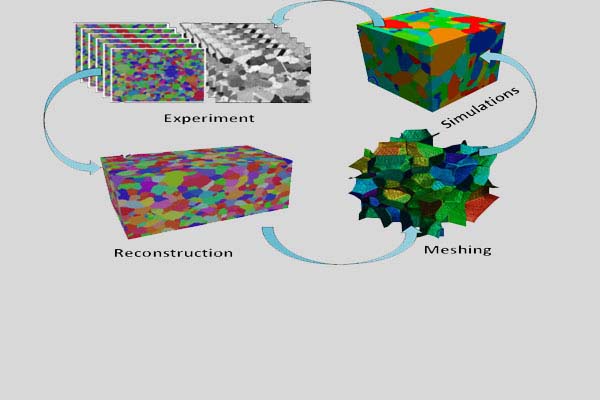
2D Materials for Water Purification
Current membranes have limited water permeability, and they deteriorate quickly. There is evidence that microporous materials like linear polymers and zeolite can achieve a variety of water fluxes with limitations such pollution rejection and selectivity. Large-scale production also has high fabrication costs. Throughout the last decade, two-dimensional (2D) materials have been used to fabricate membranes enabling the ability to produce extremely high fluxes and excellent pollutant separation. The insertion of nanopores to some 2D materials requires procedures such as plasma etching, oxidative chemical etching, ion beam irradiation, and electron beam irradiation. These methods produced high permeate flux graphene membranes with 100% rejection of sodium, lithium, potassium, and chloride ions and separation of organic particles and hexane isomers. Transition metal dichalcogenides (TMDC), like MoS2, are hydrophilic and effective in water filtration, with 99 percent salt rejection and 5-fold permeate flux. Because TMDCs are difficult to synthesize defect-free sheets, lamellar construction can be used as a filtration medium.
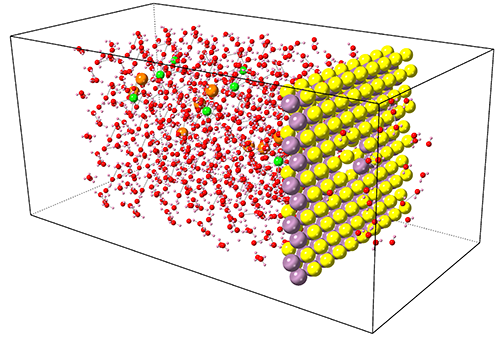
Size Effects
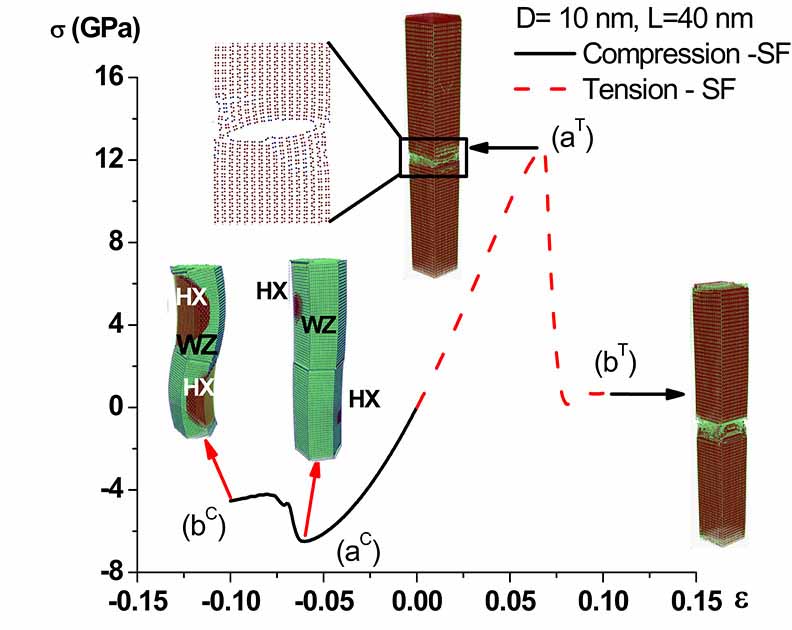
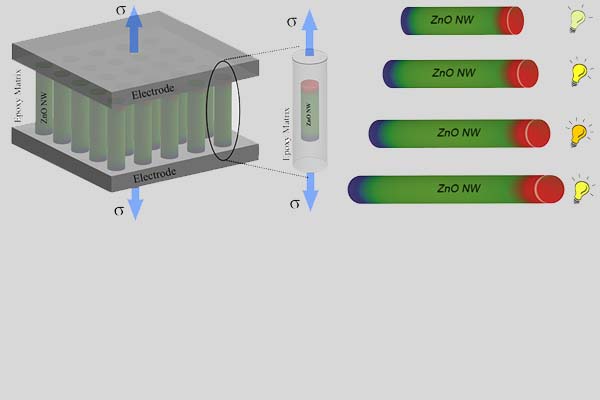
In contrast to bulk materials, in nanomaterials the number of partially-coordinated atoms with higher energy is comparable to the number of fully-coordinated low energy atoms. This is one of the key reasons behind the fascinating behavior of nanomaterials, including the size effects such as boosted electromechanical characteristics. Thus, the size of a material can be used as an additional controlling parameter for tailoring engineering material properties. We used atomistic simulations to investigate the size effects and searching for new routes for designing new materials such as materials for energy applications.
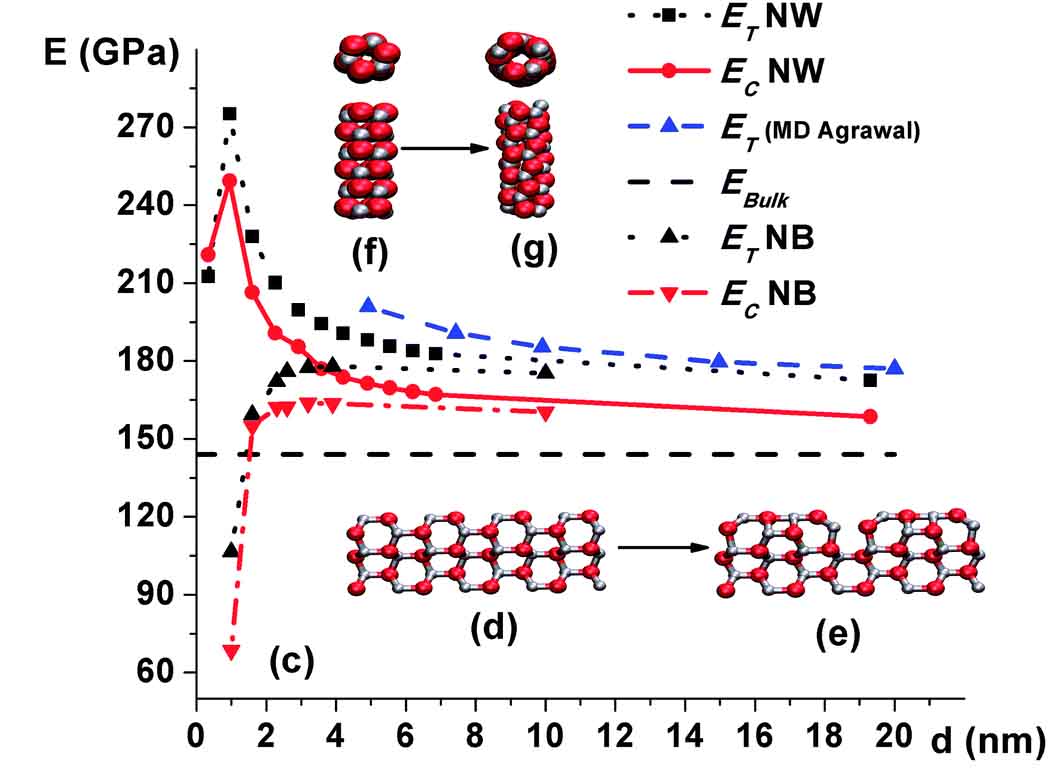
Surface Effects
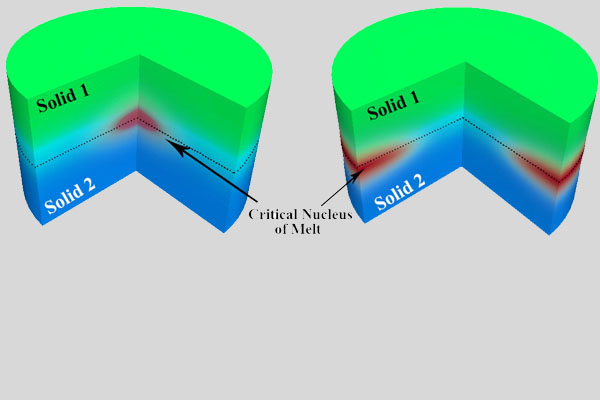
Downsizing materials to the nanoscale, where surface effects such as surface stresses dominate, is the source of surprising phenomena. Phase transformations due to these effects are known to be a path for the formation of new phases. Furthermore, we revealed that surface tension induces large stresses in the core of nanostructures, which can lead to structural instabilities and structural transformations. The newly formed structures have unique atomistic arrangements and consequently exceptional properties. In this research direction, we are seeking new ways to engineer materials by tailoring their surface properties.
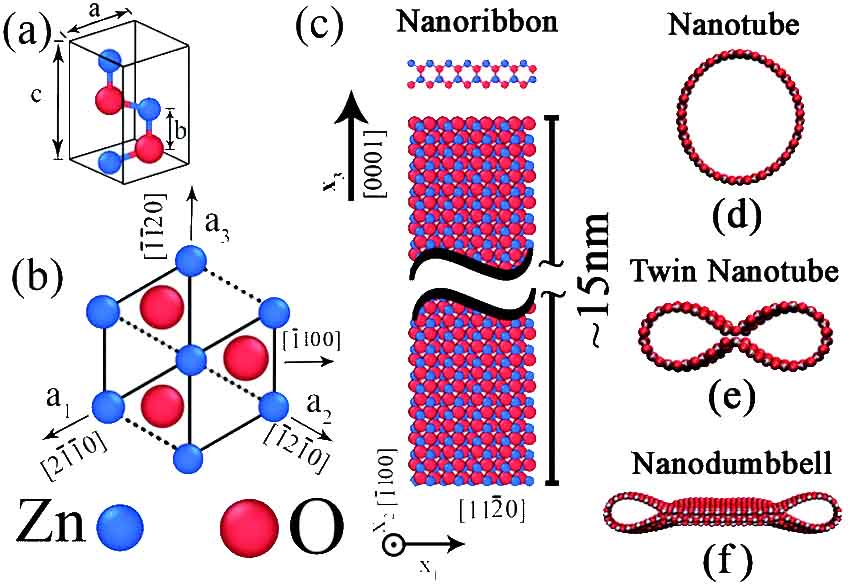
Defect Engineering
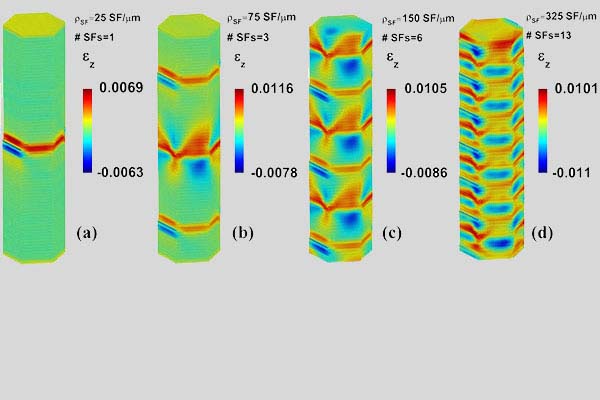
Despite our perfectionistic desires, formation of defects in real-world processes is almost inevitable, which in general degrade the mechanical properties of materials. However, it can improve other material’s characteristics such as catalytic properties. Presence of defects also increase the total energy of the material, disturbs the arrangement of atoms, and results in the formation of excess elastic energy in the system. In our research we investigate the interplay between material defects and material characteristics, and seek new ways for using defects to tailor material properties. This problem gets even more complicated in the case of nanomaterials as the defects start to interact with the external surfaces when the materials’ dimensions get smaller and smaller.